|
1/31/2024 0 Comments 30 60 90 day plan templates free 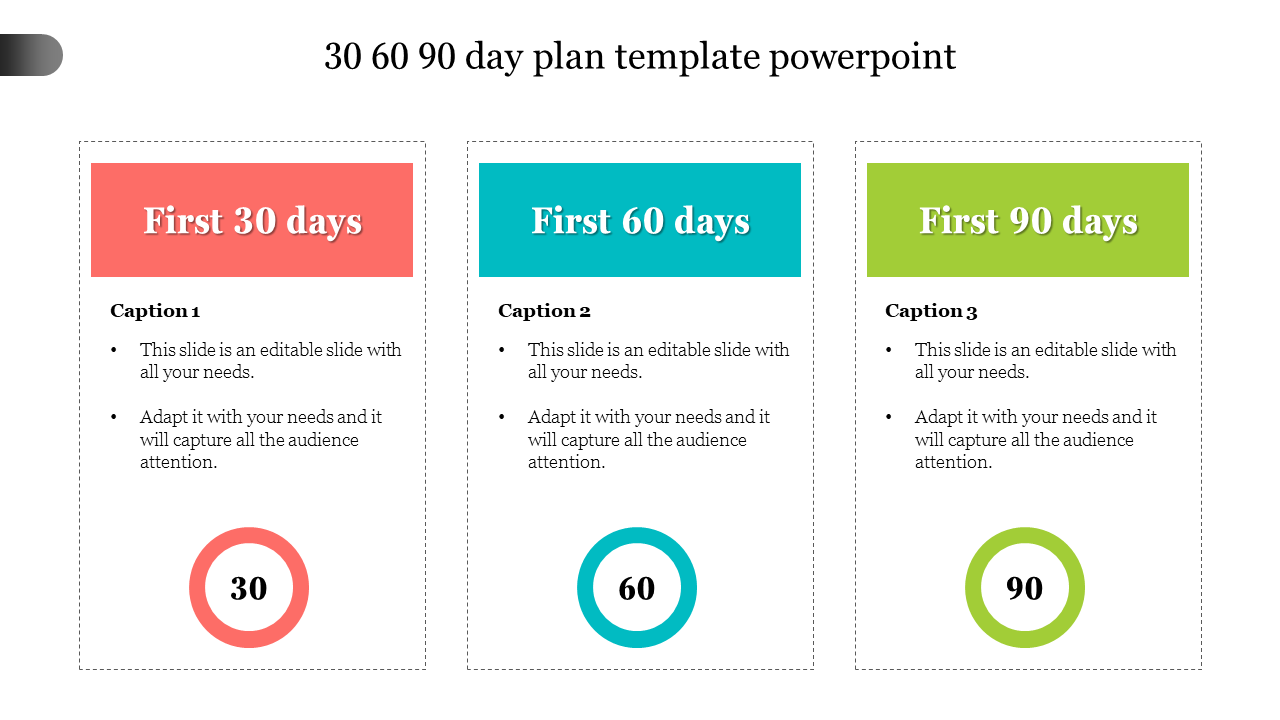
Type IA changes have little or no impact on the quality, safety or efficacy of the product, for example administrative modifications such as: These are divided into 2 types: IA and IB. Further guidance will be issued in due course. where relevant, copies of assessment reports should be provided to support the submission. If this approach is to be followed, this needs to be made clear in the application and relevant evidence, declarations and supporting information e.g. following the acceptance of the same change(s) to a related the same product as part of a European procedure e.g. Variations to purely national MAs (PL, PLGB and PLNI) can be presented to the MHRA under the reliance route i.e. Until further notice the variations classification guideline, which is a fundamental component of the operation of the variations system, will continue to apply to all types of variations. MHRA for purely national variations, the RMS for MR/DC or Reference Authority for any worksharing variations.Ĭentralised Marketing Authorisations are authorised for use in Northern Ireland only and any variations to these products will be managed by the EMA. The procedures are dependent on who is responsible for leading the assessment i.e. The variation procedures which will be followed for the different types of Marketing Authorisations from 1 January 2021 are essentially the same as before that date, where relevant, covered by the appropriate European or National legislation. as part of an MR/DC procedure including Northern Ireland only.PLNI - authorised for use in Northern Ireland only under the unfettered access route (MAH needs to be established in Northern Ireland and any product must access Great Britain, via Northern Ireland as a qualifying Northern Ireland good).following conversion of a centrally authorised product (CAP).PLGB – authorised for use in Great Britain only (England, Scotland and Wales) as part of an MR/DC procedure involving Northern Ireland as CMS, but a UK wide MA.PL – authorised for use in United Kingdom ( Guidance relating to application for a licence to market a medicine in the UK) multiple changes to one or more products and/or MAsįrom 1 January 2021, the following Market Authorisation types will be possible in the United Kingdom (UK):.a single change to one marketing authorisation (MA). 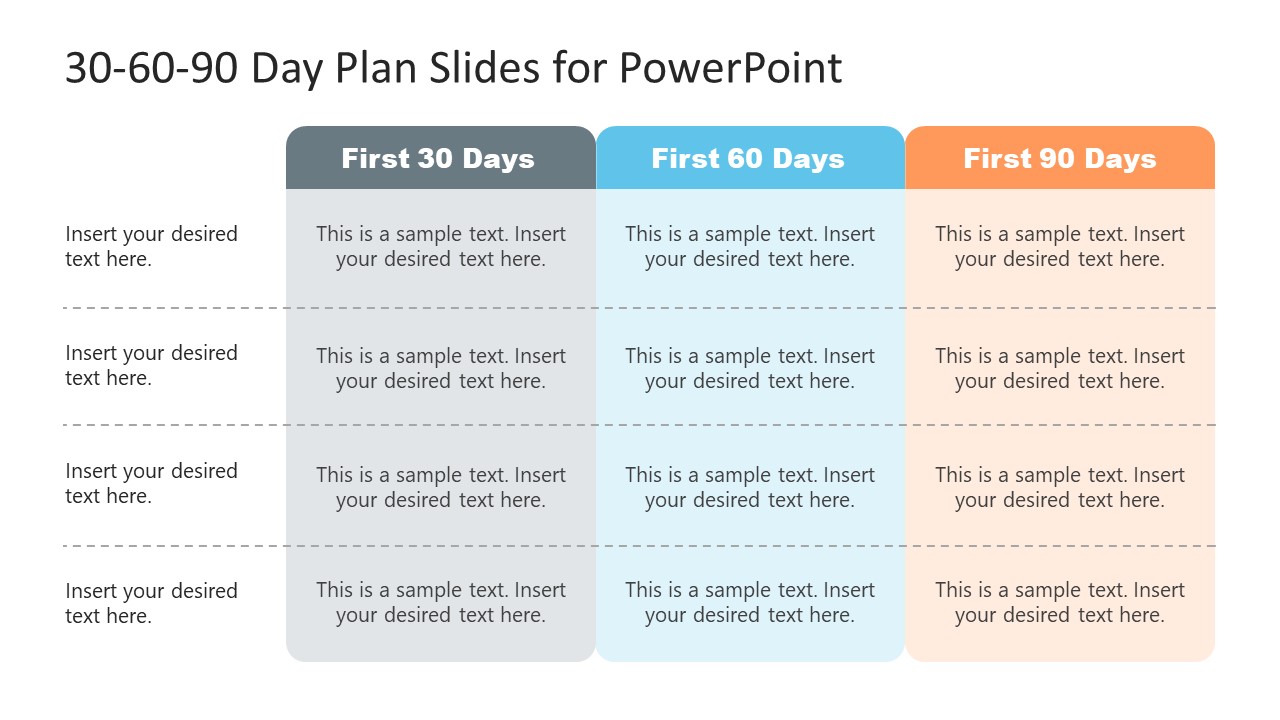
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
